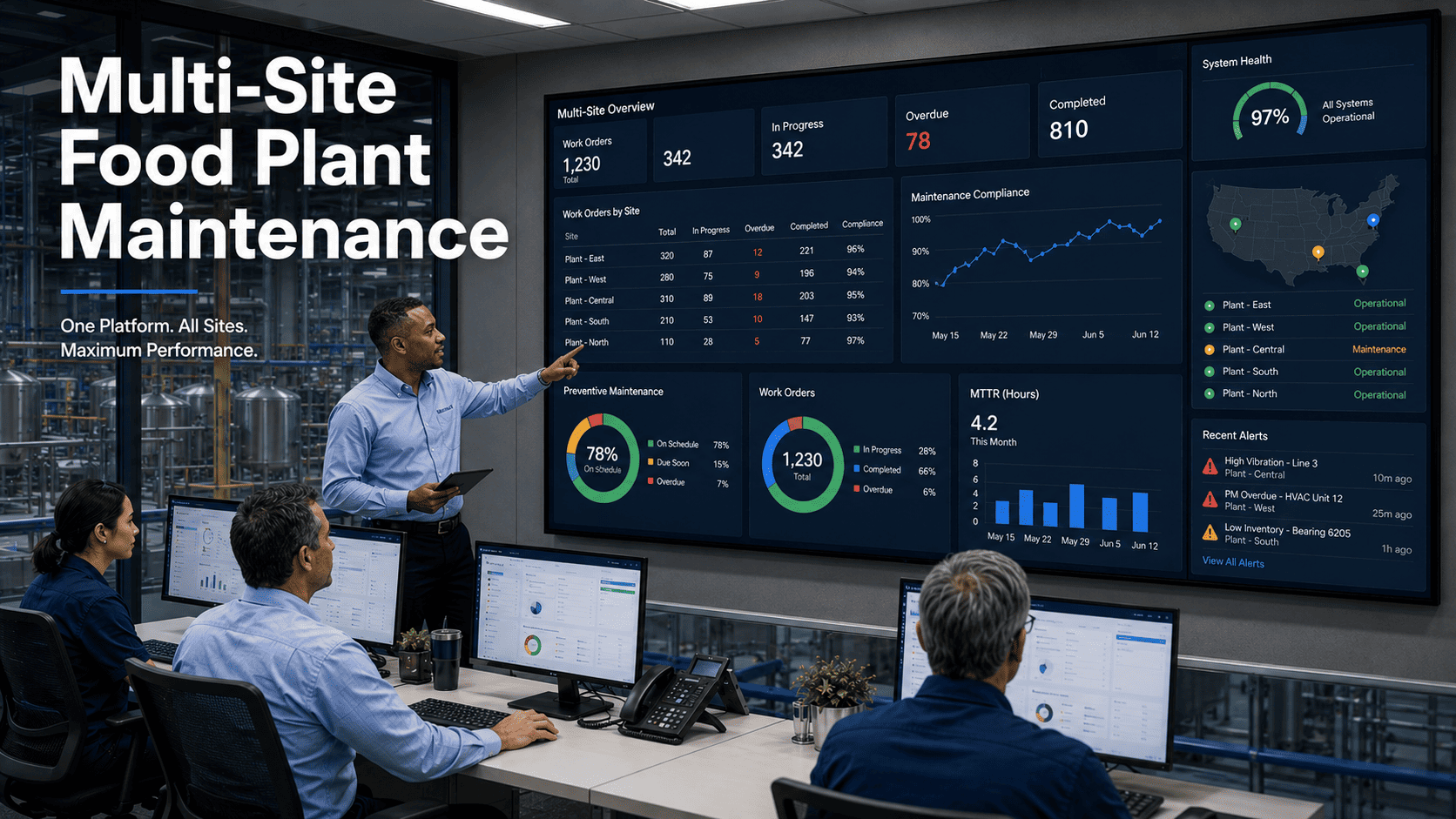
Most food plant maintenance managers believe they have real-time visibility. Their CMMS dashboard says "live." Their vendor says "real-time." But when a compressor alert fires 40 minutes after degradation began, when a closed work order still shows overdue three hours later, when three systems display three different PM completion rates for the same shift — what they actually have is the illusion of data built on a fundamentally broken maintenance pipeline. In today's food manufacturing industry, genuine maintenance visibility has the potential to prevent recalls, eliminate unplanned downtime, and ensure continuous compliance with FDA FSMA, HACCP, GMP, and SQF requirements. See what real maintenance visibility looks like in OxMaint — sign up free.
Food Manufacturing · CMMS · Compliance 2026
Preventive Maintenance and Food Safety Compliance: How CMMS Transforms Food Manufacturing Operations
FDA FSMA, HACCP, GMP, and SQF compliance all depend on documented maintenance records your team generates every single day. OxMaint's mobile-first CMMS captures every inspection, work order, calibration, and asset event automatically — turning your maintenance team into a compliance documentation engine that runs without any extra administrative burden on your staff.
$1.1T
Global food manufacturing market — maintenance failure is the leading driver of unplanned production downtime
23%
Average uptime improvement when food plants move from reactive to structured preventive maintenance programs
$2.8M
Average cost of a food product recall — 68% triggered by equipment maintenance failures preventable with proper PM
4.8x
ROI delivered by structured preventive maintenance programs in food and beverage processing operations
The Real Challenge
Why Food Manufacturing Maintenance Is Fundamentally Different From Every Other Industry
Every manufacturing plant needs maintenance. But food manufacturing carries three layers of consequence that other industries simply do not — regulatory, safety, and public health — making maintenance documentation failures qualitatively different from a missed PM on a stamping press or a delayed work order on an HVAC unit.
Consequence Level 1
Equipment Failure Becomes a Food Safety Incident
A failing temperature sensor on a pasteurization unit does not just stop production — it potentially releases under-processed product into the supply chain. A worn seal on a filling machine does not just require repair — it creates an allergen cross-contamination pathway. In food manufacturing, equipment condition is a food safety variable, not just an operational one.
68% of food recalls caused by equipment-related contamination — all preventable with structured PM
Consequence Level 2
Maintenance Records Are Regulatory Documentation
FDA FSMA, HACCP verification requirements, GMP standards, and SQF certification all require documented evidence that equipment was maintained, inspected, and calibrated on schedule. A maintenance log existing only in a supervisor's memory is an enforcement liability. Digital CMMS records are the only format that satisfies modern regulatory audit standards without hours of manual reconstruction.
FDA 483 citations for inadequate maintenance records are the fastest-growing inspection category
Consequence Level 3
Downtime Has a Perishable Goods Multiplier
An unplanned 4-hour downtime on a beverage filling line does not just cost labor and lost throughput — it potentially writes off an entire batch of perishable product in-process. A refrigeration compressor failure at 2 AM can destroy $200,000 of temperature-sensitive product before anyone discovers the fault without real-time monitoring and automated work order generation.
Food manufacturing downtime costs 3–5x the equivalent event in durable goods manufacturing
Compliance Frameworks
6 Food Safety Standards That Require Digital Maintenance Documentation
Every certification your facility holds — and every audit it will face — requires documented evidence that equipment maintenance, inspection, and calibration happened on schedule. OxMaint generates this documentation automatically from daily maintenance operations.
FDA FSMA
Food Safety Modernization Act
FSMA's Preventive Controls for Human Food rule requires written procedures for monitoring, verification, and corrective actions for every equipment-related hazard. Digital work orders and inspection records in OxMaint satisfy these requirements automatically on every transaction.
Generates: Preventive control monitoring records, corrective action docs, verification records
HACCP
Hazard Analysis Critical Control Points
HACCP CCP monitoring requires timestamped records at each critical control point — including temperature monitoring equipment, metal detectors, and sanitation checkpoints. Equipment calibration and inspection records are core HACCP documentation retrievable on demand during audits.
Generates: CCP calibration logs, inspection completion records, audit trail by technician
GMP
Good Manufacturing Practice
21 CFR Part 110/117 GMP requirements mandate documented equipment cleaning, maintenance, and calibration schedules with verification records. GMP auditors specifically test whether maintenance records can be produced within minutes — not whether they theoretically exist somewhere in a binder.
Generates: Equipment cleaning records, maintenance schedule verification, calibration certificates
SQF
Safe Quality Food Certification
SQF Level 2 and 3 certification requires a documented preventive maintenance program with scheduled frequencies, completion verification, and corrective action records for non-compliance. Plants with digital CMMS records consistently score higher than those with paper systems.
Generates: PM schedule documentation, completion rate reports, corrective action audit trail
BRC
British Retail Consortium Standard
BRC Food Safety Standard clause 4.6 requires a planned maintenance program covering all equipment with documented maintenance instructions, frequency, and completion records. BRC audits require demonstrable evidence that equipment maintenance is driving food safety outcomes.
Generates: Planned maintenance program records, equipment-specific instructions, frequency compliance
ISO 22000
Food Safety Management Systems
ISO 22000 clause 8.2 requires infrastructure maintenance as a prerequisite program with documented procedures and monitoring records. The standard requires evidence that maintenance activities are performed as planned and that deviations trigger corrective action.
Generates: Infrastructure maintenance procedures, monitoring records, deviation documentation
Root Cause Analysis
Six Reasons Maintenance Programs Break Down in Food Plants
Maintenance failure in food plants is not one problem — it is six separate failure modes that each independently degrade your production uptime and compliance posture, and that compound when they appear together. Most plants experiencing recurring downtime have four or more active simultaneously.
No Structured Preventive Maintenance Schedule
Most food plants still run maintenance reactively — fixing equipment after it fails rather than before. Without a structured PM schedule, there is no systematic process for catching developing failures during scheduled maintenance windows. A compressor beginning to degrade in February will not be discovered until it fails completely in April — by which time a $400 corrective action has become a $40,000 emergency repair with a $180,000 product loss attached. OxMaint's PM scheduling module generates work orders automatically based on time intervals, runtime hours, or production cycle counts — ensuring every critical asset receives scheduled maintenance before failure thresholds are reached.
Gap: Reactive repair costs 3–10x preventive maintenance cost · Product loss multiplier: Up to 100x per event
Paper-Based Inspection Records That Cannot Survive an FDA Audit
Paper inspection logs fail regulatory audits in three specific ways: they cannot demonstrate contemporaneous creation (FDA requires records created at the time of activity, not reconstructed later), they cannot prove technician identity without witness signatures on every form, and they cannot be retrieved in under five minutes for any equipment during an unannounced inspection. OxMaint's mobile inspection system timestamps every checklist completion at the moment of activity, attributes it to the completing technician by user account, and makes every inspection record retrievable in under 60 seconds from any device — satisfying all three FDA contemporaneous documentation requirements simultaneously.
Gap: Paper records fail FSMA contemporaneous standard · Regulatory cost: $60K–$300K per FDA finding
Maintenance, Production, and Quality Living in Separate Data Silos
Your maintenance system tracks work orders but does not know what the production schedule is. Your production system tracks output but does not know what was serviced this shift. Your quality system logs CCP readings but does not know the pasteurizer had an open work order during the same window. Without connected systems, patterns that would be immediately obvious — equipment failing during product changeovers, maintenance gaps correlating with quality holds — are completely invisible. Research shows maintenance costs run 40–60% higher when production and maintenance data stay siloed. OxMaint integrates with ERP and MES systems via live data connections, ensuring maintenance decisions are made with full production context.
Gap: Invisible cross-system failure patterns · Maintenance cost premium: 40–60% above integrated operations
Uncontrolled Vendor and Third-Party Access to Critical Equipment
Food manufacturing facilities depend on dozens of third-party vendors — equipment OEMs, sanitarians, calibration services, refrigeration contractors — each requiring access to food-contact or production-critical systems. Without digital work order control, vendor access is logged on paper or not at all: no record of what was done, what was changed, or what state the equipment was in before and after the visit. When a food safety incident investigation traces back to a vendor visit, plants without digital access logs face the worst possible position with both FDA and their customers. OxMaint's work order system enforces access scope for every vendor visit, logging every action taken and generating a complete third-party access record automatically.
Gap: Uncontrolled vendor access is an undocumented contamination risk pathway · Audit finding probability: High
No Asset Performance Visibility Until Something Breaks
Maintenance managers in plants without live asset performance dashboards discover equipment problems in one of two ways: a technician notices something during manual inspection, or production stops. Neither is acceptable in a food manufacturing environment where undetected refrigeration failure destroys product, undetected temperature drift releases out-of-spec product, and undetected conveyor wear creates a foreign material contamination risk. OxMaint's asset performance dashboard gives maintenance managers a real-time view of OEE scores, downtime frequency by asset, PM compliance rates, and open work order backlogs — providing early warning before developing failures escalate to production stoppages or food safety incidents.
Gap: Zero visibility until failure · Cold chain failure average product loss: $180K per event
SQF and BRC Recertification Consuming Weeks of Manual Record Assembly
SQF and BRC audits require presenting a documented, executed preventive maintenance program covering all equipment with frequency records, completion verification, and corrective action trails. Plants without digital CMMS spend two to four weeks before each audit cycle manually assembling PM records from paper logs, spreadsheets, and technician memory — a process that simultaneously consumes management capacity, introduces assembly errors, and creates documentation gaps that auditors flag as findings. OxMaint generates SQF and BRC audit-ready compliance reports directly from daily maintenance operations, with zero additional administrative effort. When the audit arrives, the documentation is already complete.
Gap: 2–4 weeks of manual record assembly per audit cycle · SQF/BRC finding rate from paper systems: 3x digital
OxMaint closes all six failure modes in a single platform
Digital inspections. Automated PM scheduling. Audit-ready compliance records. Live in 14 days — no IT project required.
Equipment Coverage
Every Critical Equipment Type in Your Food Plant — Covered by OxMaint's Digital Inspection and PM System
OxMaint's customizable inspection checklists and preventive maintenance schedules cover every equipment category in food and beverage manufacturing — from high-speed packaging lines to cold-chain refrigeration to CIP sanitation systems.
Processing Equipment
Mixers and blenders — bearing, seal, and blade inspection schedules
Pasteurizers — temperature sensor calibration and flow rate verification
Homogenizers — pressure system and valve PM schedules
Evaporators and dryers — heat transfer surface and moisture control checks
Pasteurizer temperature sensor drift responsible for 34% of dairy recall events — preventable with scheduled calibration
Packaging and Filling Lines
Filling machines — seal integrity and nozzle condition inspection cycles
Labeling systems — registration accuracy and adhesive system PM
Coding and dating equipment — print quality verification and ink system checks
Conveyor systems — belt tension, bearing condition, and guide rail inspection
Filling machine seal failures are the #1 cause of allergen cross-contamination on multi-product lines
Cold Chain and Refrigeration
Refrigeration compressors — pressure differential checks and refrigerant level PM
Cold storage units — temperature sensor calibration and door seal inspection
Blast chillers — airflow and temperature uniformity verification
Condenser coils — fouling and cleaning schedule management
Undetected refrigeration failure destroys an average of $180,000 in product per event at mid-size food plants
Sanitation and CIP Systems
CIP systems — chemical concentration verification and spray nozzle coverage checks
Sanitation equipment — brush condition and chemical dispenser calibration
Drain and floor systems — slope, seal, and trap inspection for pest exclusion
Air handling units — filter condition and positive pressure verification
CIP system failures account for 41% of microbiological contamination events in dairy and beverage plants
How OxMaint Works
Point-by-Point: How OxMaint Closes Each Maintenance Failure Mode
OxMaint is built as a mobile-first operational platform — not a desktop system with a mobile afterthought. Each capability below directly addresses a specific failure mode that causes unplanned downtime or compliance exposure in food manufacturing operations.
No structured PM schedule (Failure 01)
→
Automated PM scheduling by time interval, runtime hours, or production cycle counts. Work orders generated before failure thresholds. Plants report 23% average uptime improvement within 90 days of deployment.
Paper inspection records (Failure 02)
→
Mobile digital checklists timestamped at moment of activity, attributed to named technician by user account. Every inspection retrievable in under 60 seconds during any FDA, HACCP, or SQF audit.
Siloed maintenance and production data (Failure 03)
→
Open API integration with ERP and MES systems. Production schedule context feeds maintenance decision-making. Failure patterns invisible in siloed systems become immediately visible in OxMaint's unified dashboard.
Uncontrolled vendor access (Failure 04)
→
Digital work orders enforce access scope for every vendor visit. Every action logged, every equipment state before and after captured. Complete third-party access record generated automatically for each visit.
No asset performance visibility (Failure 05)
→
Real-time asset performance dashboard: OEE scores, downtime frequency by asset, PM compliance rates, open work order backlogs. Automated work orders generated when performance metrics cross alert thresholds.
Manual SQF/BRC audit record assembly (Failure 06)
→
SQF and BRC audit-ready compliance reports generated directly from daily maintenance operations. Zero additional administrative effort. When the audit arrives, the documentation is already complete — no 2–4 week assembly sprint required.
Performance Data
What Food Manufacturing Plants Measure After Deploying OxMaint
23%
Uptime Improvement
Average production uptime improvement within 90 days of deploying structured PM schedules — driven by catching failures before they cause unplanned stoppages.
68%
Fewer Unplanned Stoppages
Reduction in unplanned equipment stoppages when plants transition from reactive to preventive maintenance supported by digital inspection and work order management.
4.8x
Maintenance ROI
Return on maintenance investment delivered by structured PM programs — driven by avoided downtime costs, extended equipment life, and reduced emergency repair spend.
60 sec
Audit Record Retrieval
Time to retrieve complete maintenance and inspection history for any equipment during FDA or food safety audit — vs. 4–8 hours of manual record assembly from paper systems.
$2.8M
Average Recall Cost Avoided
Average cost of a food product recall — 68% triggered by equipment maintenance failures that structured PM programs with digital inspection records directly prevent.
14 days
Go-Live Timeline
From sign-up to fully operational OxMaint deployment across your food manufacturing facility — cloud-based, mobile-first, no IT infrastructure project required.
Self-Assessment Tool
Is Your Food Plant Maintenance Program Compliance-Ready? 8 Diagnostic Questions
These questions diagnose whether your current maintenance program can withstand an unannounced FDA inspection, an SQF audit, or a recall investigation — each targets a specific documentation or operational capability you can verify right now.
Can you retrieve complete maintenance records for any asset for any 90-day period in under 5 minutes?
Testing: Audit-ready digital records vs. paper binders requiring manual assembly
If no: Your records are not audit-ready. FDA visits do not wait for record assembly.
Does every inspection record show a timestamp at the moment of completion and the name of the completing technician?
Testing: Contemporaneous documentation vs. retrospective batch records
If no: Your records fail FSMA contemporaneous documentation standard.
Are PM work orders generated automatically before equipment reaches failure thresholds — or does someone have to remember to create them?
Testing: Automated PM scheduling vs. calendar reminders and supervisor memory
If no: Your PM program depends on memory. PMs will be missed under production pressure.
Is there a complete digital log of every vendor access to food-contact or production-critical equipment in the last 12 months?
Testing: Controlled vendor access with work order records vs. unlogged third-party visits
If no: Vendor access is an undocumented contamination risk pathway in your facility.
Can you generate a complete SQF or BRC compliance report covering all PM activities in the last 12 months right now — without assembling records from multiple sources?
Testing: Integrated compliance reporting vs. manual multi-source record assembly
If no: Your next SQF/BRC audit will require 2–4 weeks of manual preparation effort.
Do you have a live view of PM completion rates, overdue work orders, and equipment performance metrics at this moment?
Testing: Live maintenance performance dashboard vs. end-of-shift reports or spreadsheets
If no: Maintenance decisions are being made without current performance context.
When a technician completes a work order in a refrigerated room with no WiFi, does it appear complete on your system within 2 minutes?
Testing: Offline-capable mobile sync vs. connectivity-dependent work order recording
If no: Work orders in connectivity-poor areas queue and create phantom compliance gaps for hours.
If a recall investigation occurred today, could you confirm the maintenance condition of every production-critical asset at the time of affected lot production?
Testing: Complete timestamped asset maintenance history vs. fragmented records
If no: A recall investigation will find documentation gaps that expand recall scope and FDA exposure.
0–2 yesCritical compliance and operational exposure. Structural documentation failures in multiple categories. Immediate action required before next FDA inspection or audit cycle.
3–5 yesModerate gap. Some processes in place but significant documentation weaknesses that will generate findings during a rigorous SQF, BRC, or FDA audit.
6–8 yesStrong foundation. Minor remaining gaps. OxMaint can close the final ones and add real-time asset performance visibility across your full facility.
Detailed FAQ
Food Manufacturing CMMS — Questions Answered in Full
What is a CMMS and why do food manufacturing plants specifically need one?
A CMMS (Computerized Maintenance Management System) is a digital platform that manages all maintenance activities for a facility — work orders, preventive maintenance schedules, equipment inspection records, spare parts inventory, and compliance documentation. Food manufacturing plants have a specific, non-negotiable need for CMMS that other industries do not share: regulatory documentation. FDA FSMA, HACCP verification, GMP compliance, and SQF certification all require written evidence that equipment was maintained, inspected, and calibrated on schedule — and that deviations triggered corrective action. A paper-based maintenance system cannot produce this documentation at audit standard during an unannounced FDA inspection. OxMaint's digital CMMS generates FDA, HACCP, GMP, SQF, BRC, and ISO 22000-compliant documentation automatically from daily maintenance operations.
Sign up free to see the compliance documentation module, or
book a live demo for a guided walk-through.
How does OxMaint help food plants pass FDA inspections and HACCP audits?
OxMaint helps food plants pass FDA inspections and HACCP audits through four specific documentation mechanisms that regulators look for. First, equipment-specific inspection records: every inspection completed in OxMaint is timestamped, attributed to a named technician, and linked to the equipment record — satisfying FDA's requirement for documented monitoring procedures. Second, preventive maintenance completion records: OxMaint tracks PM schedule adherence and generates completion reports demonstrating your PM program is actually being executed. Third, corrective action trails: when an inspection fails or a PM reveals a deficiency, OxMaint automatically generates a corrective action work order linked to the original inspection record. Fourth, calibration records: temperature sensors and measurement equipment at critical control points are managed in OxMaint with calibration schedules and out-of-calibration event documentation. During an unannounced FDA visit, OxMaint users produce complete equipment histories in under 60 seconds.
Sign up free to configure your HACCP documentation workflow.
How quickly does OxMaint deploy in a food manufacturing facility?
OxMaint deploys in 7–14 days for a standard food manufacturing facility. Phase 1 — Asset Setup (Days 1–3): OxMaint's onboarding team works with your maintenance manager to upload your equipment registry, asset hierarchy, and existing PM schedule frequencies. This requires approximately 4–6 hours of your team's time. Phase 2 — Checklist and Schedule Configuration (Days 4–7): OxMaint's food manufacturing checklist library provides pre-built inspection templates that your team can customize to match your specific equipment models and compliance requirements. Phase 3 — Mobile Training and Go-Live (Days 8–14): Maintenance technicians and supervisors complete mobile app training in 2–3 hours. Because OxMaint is cloud-based and mobile-first, there is no server infrastructure to install, no IT department project, and no hardware procurement for the core deployment.
Sign up free to start the deployment process, or
book a demo for a deployment timeline walkthrough specific to your facility.
Can OxMaint handle multi-site food manufacturing operations with different compliance requirements at each facility?
OxMaint is built as a multi-site platform from its core architecture — a single deployment covers all facilities in your food manufacturing portfolio under one unified dashboard with site-specific configurations maintained independently per location. A bakery facility requiring BRC Level 2, a dairy processing plant requiring HACCP and FDA 21 CFR Part 117 records, and a beverage bottling plant requiring SQF Level 3 certification can all run simultaneously under the same OxMaint agreement. At the corporate level, operations directors see KPI comparisons across all sites — PM compliance rates, inspection completion percentages, open work order backlogs, and OEE trends — enabling performance benchmarking across the entire manufacturing portfolio. OxMaint supports regulatory documentation frameworks for FDA, EFSA, FSA, FSSAI, and FSANZ from the same platform.
Book a demo to see multi-site food manufacturing configuration running live.
What is the ROI of deploying OxMaint in a food and beverage manufacturing plant?
The ROI calculation has five independent financial components. Unplanned downtime prevention: food plants using OxMaint report a 23% average uptime improvement within 90 days. For a plant with $50,000/hour of production value, preventing even one 4-hour unplanned shutdown per month generates $2.4M in annual production protection. Recall cost avoidance: the average food product recall costs $2.8M. 68% of recalls are caused by equipment-related contamination pathways — all preventable with documented PM programs. Regulatory penalty avoidance: FDA warning letters triggered by inadequate maintenance documentation can impose corrective action costs of $500,000–$5M per enforcement action. SQF and BRC certification: facilities with digital CMMS consistently achieve higher scores and smoother recertification than paper-based competitors. Emergency repair reduction: plants on structured PM programs spend 25–35% less on emergency repairs. OxMaint customers typically achieve full platform payback within 3–6 months of go-live.
Sign up free to start measuring ROI from day one.
We already have an existing CMMS. Is switching to OxMaint worth the disruption?
This question requires honest analysis of two numbers: the sunk cost of your existing investment, and the ongoing cost of the compliance and operational gaps it creates. The ongoing cost of the six failure modes described on this page — typically $335K–$1.49M annually for a mid-size food plant — is real and accruing every shift. OxMaint's onboarding team can import your historical maintenance data from most common CMMS formats, preserving your complete asset maintenance history while replacing the structural limitations constraining your current platform. Your past data comes with you. The structural problems do not.
Book a demo to walk through an ROI analysis specific to your operation.
Every Gap in Your Maintenance Records Is a Gap in Your Food Safety Defense
Real Compliance Documentation Is Not a Byproduct of Good Intentions — It Is a Byproduct of the Right System Running Every Shift.
OxMaint gives food manufacturing operations the digital inspection tools, preventive maintenance scheduling, work order management, and compliance documentation that FDA, HACCP, GMP, SQF, and BRC auditors require — generated automatically as a byproduct of daily maintenance operations. Start free today. No IT project. No hardware. Live in 14 days.
60 sec
Audit record retrieval