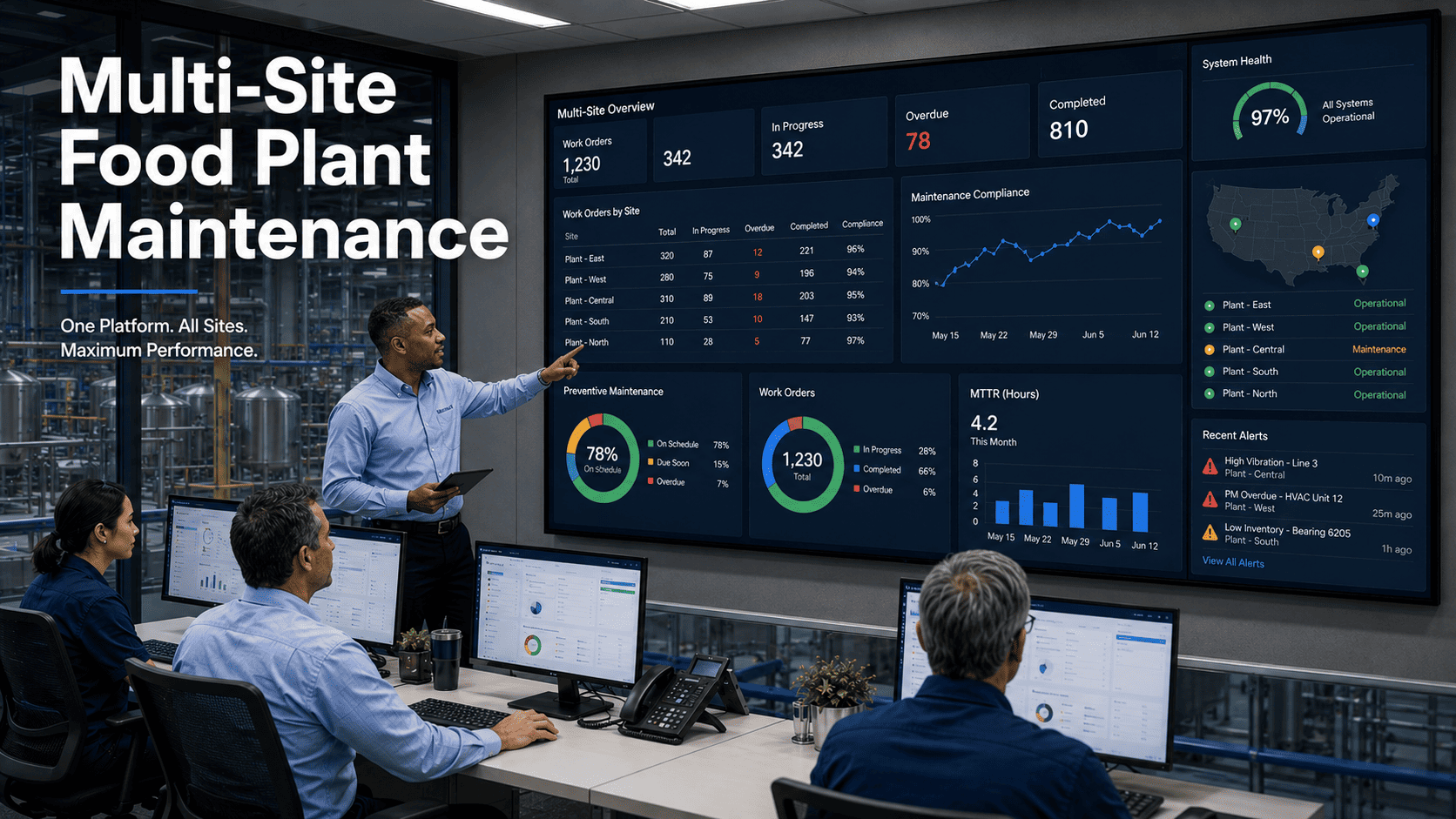
A dairy processor in Wisconsin discovered their CIP system had been underperforming for months—spray balls were clogged, pump seals were leaking, and chemical concentrations had drifted from specifications. The result: a Listeria-positive environmental swab that triggered a voluntary recall, FDA scrutiny, and $2.3 million in losses. Their CIP equipment had been "running" but nobody was systematically verifying it was actually cleaning. Facilities using by Signing up on Oxmaint for sanitation maintenance tracking catch these degradation patterns before they become food safety incidents.
Clean-in-Place systems are the backbone of food safety in processing facilities—automating the cleaning of tanks, piping, and equipment without disassembly. But CIP effectiveness depends entirely on proper maintenance. Worn spray balls don't deliver coverage. Degraded pump performance reduces flow rates. Faulty sensors approve inadequate cycles. This guide covers the maintenance practices that keep CIP systems performing to specification. Book a demo to see how Oxmaint's Sanitation Maintenance Tracking ensures your CIP equipment stays audit-ready.
Maintenance Guide / Sanitation & Hygiene
CIP System Maintenance for Food & Beverage Plants
Essential maintenance practices to ensure Clean-in-Place systems deliver consistent sanitation and regulatory compliance.
4Critical Parameters
TACTTime, Action, Chemistry, Temp
100%Coverage Required
DailyVerification Needed
Understanding CIP System Components
Effective CIP maintenance requires understanding each component's function and failure modes. Every element in the system affects cleaning performance.
PMP
Supply & Return Pumps
Deliver cleaning solutions at specified flow rates and pressures. Centrifugal pumps are standard; positive displacement for high-viscosity applications.
Critical: Flow rate determines mechanical action on surfaces
SPR
Spray Devices
Spray balls, rotating jets, and static devices distribute solutions across tank surfaces. Pattern and coverage depend on device condition and positioning.
Critical: Clogged or worn devices create coverage gaps
HTX
Heat Exchangers
Maintain solution temperatures throughout cleaning cycles. Plate or shell-and-tube designs; temperature consistency affects chemical efficacy.
Critical: Fouled exchangers reduce heating capacity
CHM
Chemical Dosing
Metering pumps and injection systems maintain proper concentrations. Includes caustic, acid, and sanitizer dosing systems with inline conductivity monitoring.
Critical: Wrong concentration = inadequate or unsafe cleaning
VLV
Valves & Piping
Automated valves route solutions through circuits. Mix-proof valves prevent cross-contamination between product and CIP solutions.
Critical: Valve failures can contaminate product
SNS
Sensors & Controls
Temperature, conductivity, flow, and turbidity sensors verify cycle parameters. PLC controls sequence operations and log data.
Critical: Faulty sensors approve bad cycles
Track Every CIP Component
Oxmaint's Sanitation Maintenance Tracking links CIP equipment to PM schedules, verification records, and compliance documentation.
Daily CIP System Verification
Verify these parameters every production day. Deviations require immediate investigation before approving equipment for production. Sign Up and digitize daily checks with Oxmaint.
Wash temperaturePer SOP (typically 60-85°C)
Rinse temperaturePer SOP specification
Final rinse tempAmbient or specified
Caustic concentrationTitration or conductivity
Acid concentrationTitration or conductivity
Sanitizer levelTest strips or sensor
Supply flow ratePer circuit specification
Return flow rateWithin 10% of supply
Supply pressurePer spray device rating
Pre-rinse durationPer validated SOP
Wash durationPer validated SOP
Final rinse durationUntil conductivity clears
Weekly Maintenance Tasks
Schedule weekly CIP maintenance during production breaks. Document all findings and corrective actions.
1
Spray Ball Inspection
Remove and visually inspect spray balls for clogging, wear, or damage. Check spray pattern with water test. Replace any with blocked holes or uneven patterns.
2
Pump Seal Check
Inspect mechanical seals for leakage. Check seal flush systems if installed. Minor weeping indicates seal wear requiring planned replacement.
3
Strainer Cleaning
Clean all line strainers and filters. Inspect baskets for damage. Document debris type—excessive buildup indicates upstream issues.
4
Valve Operation Test
Cycle all automated valves through full range. Listen for abnormal sounds. Check actuators for air leaks. Verify position feedback signals.
5
Sensor Verification
Compare CIP sensor readings against portable reference instruments. Document any deviations exceeding calibration tolerances.
Monthly & Quarterly Service
These tasks require extended downtime and may need specialized technicians. Schedule a consultation to build your CIP maintenance calendar.
Heat exchanger inspection and cleaning
Chemical dosing pump calibration
Gasket inspection on all connections
Tank spray coverage verification
Conductivity sensor calibration
Pump performance testing (flow/pressure curves)
Mix-proof valve inspection and seat replacement
Temperature sensor calibration
PLC program backup and review
Complete circuit flow verification
Full CIP validation study
Heat exchanger disassembly and inspection
Pump rebuild or replacement assessment
Piping inspection (borescope if needed)
Control system software updates
Automate CIP Maintenance Scheduling
Oxmaint generates PM tasks automatically, tracks completion, and maintains the documentation auditors expect.
Common CIP Failures & Solutions
Recognize these issues early to prevent sanitation failures and production delays.
Inadequate cleaning
Residue on surfaces, failed ATP swabs
Low flow, wrong chemistry, short time
Verify TACT parameters, check spray coverage
Extended cycle times
Cycles run longer than validated
Fouled sensors, reduced flow, worn pumps
Calibrate sensors, inspect pumps
Chemical carry-over
High conductivity in final rinse
Insufficient rinse volume, dead legs
Extend rinse, verify piping design
Temperature drops
Wash temp falls below setpoint
Fouled heat exchanger, steam issues
Clean exchanger, check steam supply
Flow rate decline
Lower than baseline flow readings
Pump wear, clogged strainers, valve issues
Clean strainers, test pump performance
Documentation Requirements
Maintain these records for regulatory compliance and validation evidence. Sign Up and use Oxmaint to store records digitally with automatic backup.
Cycle Records
Time, temperature, flow, and concentration data for each CIP cycle with pass/fail status.
Maintenance Logs
PM completions, repairs, parts replaced, and technician notes with timestamps.
Calibration Records
Sensor calibration dates, standards used, before/after readings, and next due dates.
Validation Studies
Initial and periodic validation reports proving CIP cycles achieve required cleanliness.
Frequently Asked Questions
How often should CIP systems be validated?
Initial validation is required before use, with revalidation after significant changes (new products, modified cycles, equipment changes). Many facilities conduct annual revalidation as best practice.
Book a consultation to review your validation schedule.
What are the TACT parameters in CIP?
TACT stands for Time (cycle duration), Action (mechanical force from flow), Chemistry (cleaning agent concentration), and Temperature. All four must be within specification for effective cleaning—weakness in one can sometimes be offset by increasing another.
How do I know if spray balls need replacement?
Can CIP maintenance records satisfy auditors?
Yes—auditors (SQF, BRC, FDA) expect documented evidence that CIP equipment is maintained and verified. Digital records with timestamps, automatic calibration reminders, and searchable history satisfy audit requirements more reliably than paper logs.
Keep Your CIP Systems Audit-Ready
Oxmaint helps food manufacturers maintain CIP equipment with scheduled PMs, verification tracking, and compliance documentation.