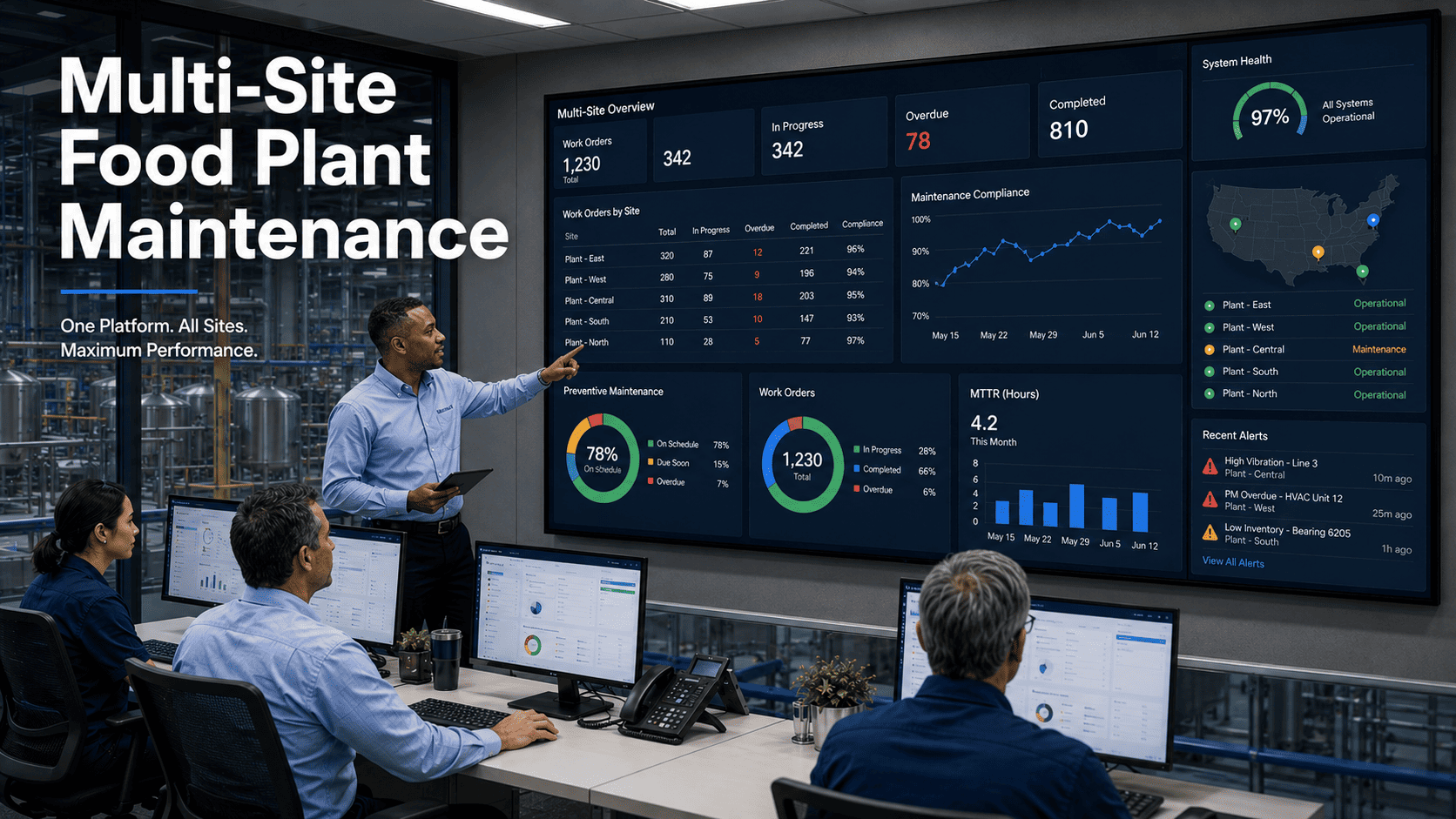
A mid-size frozen food manufacturer in Ohio lost $4.2 million in a single week—not from a fire, not from a flood, but from a metal fragment that broke off an unmaintained conveyor blade and ended up in 38,000 units of packaged chicken. The USDA classified it as a Class I recall. The root cause? A preventive maintenance task that was overdue by 47 days. According to industry benchmarks, equipment failure is cited by nearly 50% of food manufacturers as their most common source of foreign material contamination. The connection between poor maintenance management and recall risk isn't theoretical—it's costing the food industry billions every year. Sign up for Oxmaint to build a predictive maintenance system that catches contamination risks before they become recalls.
How Poor Maintenance Management Increases Recall & Contamination Risk
Food recalls in the U.S. have surged dramatically, with hospitalizations and deaths doubling in recent years. Behind many of these headlines lies a hidden culprit: inadequate equipment maintenance. Worn blades, cracked seals, and skipped inspections quietly turn production lines into contamination risks.
The Anatomy of a Maintenance-Related Recall
Most contamination events don't happen overnight. They follow a predictable chain of failures that a structured maintenance program would catch. Here's how a single skipped PM task escalates into a multi-million dollar crisis.
Wear Begins Unnoticed
A conveyor blade, gasket, or screen develops micro-cracks from continuous use. No scheduled inspection catches it.
Component Degrades Further
Vibration loosens bolts. Plastic guards become brittle. Metal fatigue worsens. The PM task is still overdue in a paper log somewhere.
Fragment Breaks Off Into Product
A metal shard, plastic chip, or rubber piece enters the product stream. Detection equipment may or may not catch it depending on material and size.
Contaminated Product Ships
Thousands of units reach retailers. A consumer complaint or regulatory inspection triggers investigation. Recall is issued. Brand trust collapses.
The 5 Maintenance Failures That Trigger Recalls
Not all maintenance gaps carry equal risk. These are the five most dangerous failure patterns in food manufacturing—ranked by how frequently they lead to contamination events and recalls.
Skipped Preventive Maintenance
When PM schedules slip, worn components like blades, seals, and screens stay in service past safe limits. Metal shards and plastic fragments enter the product stream undetected.
Sanitation Equipment Neglect
CIP systems, sanitizer dispensers, and drain infrastructure that aren't maintained create harborage points where Listeria, Salmonella, and E. coli thrive and spread to food-contact surfaces.
Failure to Track Repair Parts
Maintenance repairs themselves introduce risk. Loose bolts, broken tool tips, and replacement gasket trimmings left inside equipment become contaminants in the next production run.
Missing Documentation & Audit Trails
Paper-based logs and spreadsheets make it impossible to prove maintenance compliance during FDA/USDA inspections. Gaps in records trigger deeper investigations and enforcement actions.
No Root-Cause Tracking
Without linking incidents to equipment history, the same failure repeats. A bearing that fails every 90 days keeps contaminating product because no one connects the maintenance dots.
Reactive Maintenance vs. Predictive CMMS Approach
The gap between "fix it when it breaks" and "prevent it before it fails" is often the gap between a smooth audit and a catastrophic recall. Sign up for Oxmaint to make the shift.
Reactive / Paper-Based
"We'll fix it when it breaks"
Oxmaint CMMS
"Prevent failures before contamination"
How Oxmaint Reduces Contamination & Recall Risk
Purpose-built maintenance management tools that address the exact failure points food manufacturers face every day. Schedule a demo to see these in action.
Automated PM Scheduling
Never miss a preventive maintenance task. Set time-based or meter-based triggers that auto-generate work orders for critical food-contact equipment.
Inspection Checklists
Digital checklists for pre-production, mid-shift, and post-sanitation inspections. Photo documentation captures equipment condition in real time.
Equipment History & Trends
Track every repair, replacement, and incident per asset. Spot recurring failures and connect maintenance gaps to contamination events.
Real-Time Alerts & Escalation
Instant notifications when PM tasks go overdue, when equipment readings exceed thresholds, or when inspections flag anomalies.
Audit-Ready Reports
Generate complete maintenance compliance documentation for FDA, USDA, SQF, and BRC audits with a single click. No more paper scrambles.
Root-Cause Analysis
Link every incident back to asset data, maintenance history, and work orders. Break the cycle of recurring contamination from the same equipment.
Frequently Asked Questions
Your Next Recall Is Preventable
Every day without structured maintenance management is another day equipment silently degrades toward contamination. Don't wait for a consumer complaint or FDA investigation to expose the gap. Take control now.