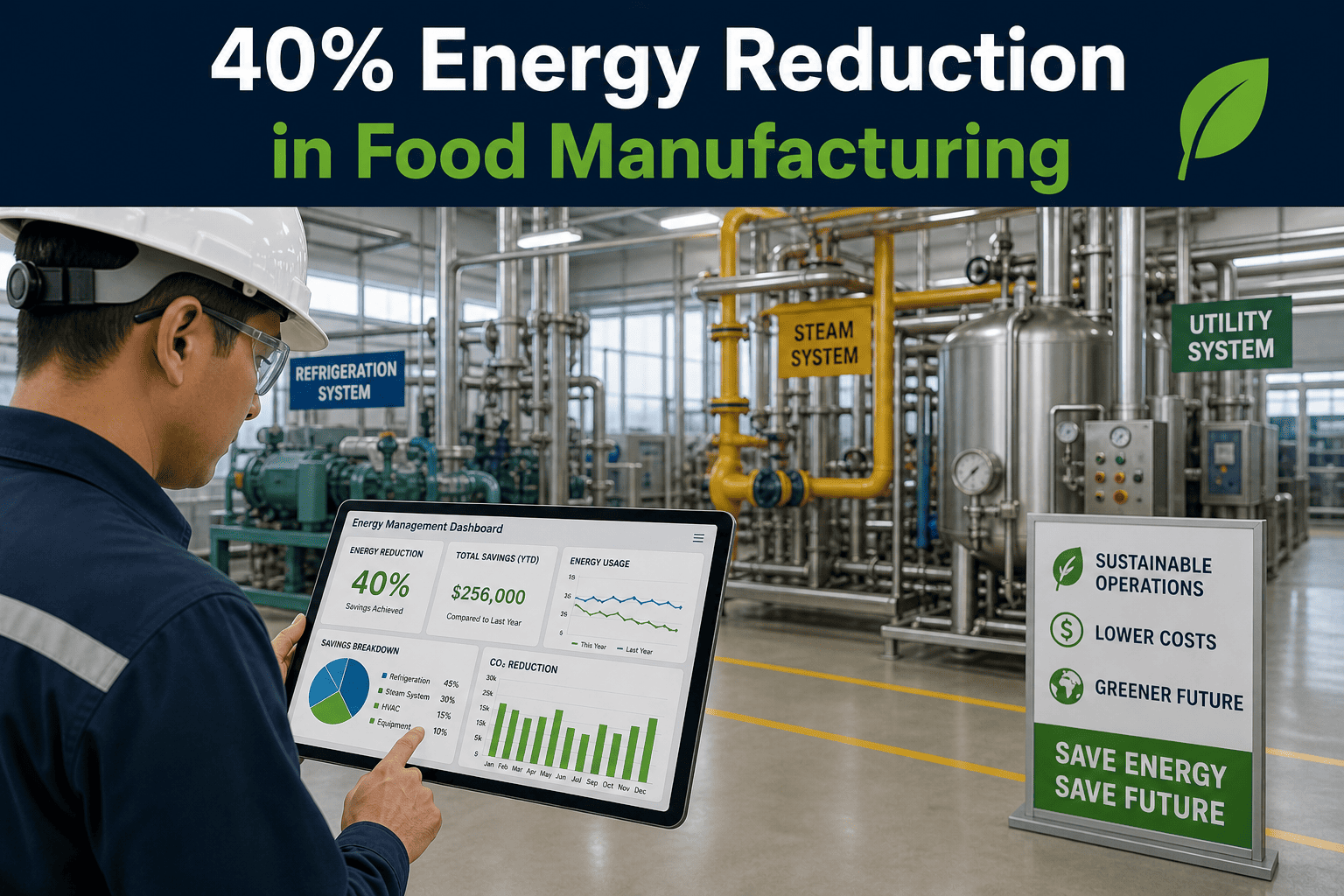
Most confectionery plants don't fail BRC audits because of bad intentions — they fail because maintenance records are scattered, calibration logs are incomplete, and allergen changeover documentation is buried in paper binders nobody can find during the audit. A single unverified CCP check or a missing preventive maintenance record can drop a Grade AA facility to a B in minutes. Industry data shows that 68% of BRC major non-conformances in food manufacturing trace back to documentation gaps — not actual safety failures. This case study covers how one mid-sized candy manufacturer restructured their entire maintenance and compliance workflow using Oxmaint CMMS, achieved BRC Grade AA on their next audit, and eliminated the pre-audit scramble that cost their team two weeks of overtime every year. Start a free trial to see how Oxmaint handles BRC documentation, or book a demo to walk through a live candy plant compliance setup.
Case Study · Confectionery · BRC Certification
How a Candy Manufacturer Achieved BRC Grade AA Using CMMS
From audit anxiety to Grade AA certification — a real-world look at how preventive maintenance, calibration tracking, and allergen changeover documentation transformed compliance for a mid-sized confectionery plant.
BRC AA
Certification achieved on first post-CMMS audit
68%
Of BRC non-conformances tied to documentation gaps, not safety failures
2 weeks
Of pre-audit overtime eliminated with digital maintenance records
100%
Calibration certificate compliance maintained across all CCP monitoring devices
Background
The Plant Before CMMS: What BRC Auditors Were Finding
This manufacturer produces hard candy, gummies, and chocolate-coated confections across a 60,000 sq ft facility with eight production lines. They had held BRC certification for several years but consistently scored in the B range — never achieving the Grade AA status required by their largest retail customer.
Their maintenance team used a combination of paper work orders, shared spreadsheets, and a legacy scheduling system that hadn't been updated in four years. Calibration due dates lived in a spreadsheet owned by one person. Allergen changeover records were handwritten and stored in a binder at each line.
When their retail buyer issued an ultimatum — Grade AA within 12 months or lose preferred supplier status — the plant manager knew the answer wasn't more procedures. It was better systems. They chose Oxmaint. Start a free trial and see how easy the transition is, or book a demo to walk through a setup built for food manufacturing compliance.
Non-Conformances from Previous Audits
Calibration certificate for two CCP thermometers found expired by 4 months
Allergen changeover log incomplete on Line 3 — missing supervisor sign-off for 11 of 30 runs
Preventive maintenance for metal detector sensitivity check — 6-week gap in records
No documented corrective action for a rejected product batch 3 months prior
Equipment cleaning verification records not linked to production batch records
Pest control equipment inspection log — no evidence of monthly inspection for Q2
Root Cause
Why Candy Plants Struggle With BRC Documentation
01
Calibration Management Is Invisible Until Audit Day
Confectionery plants use dozens of calibrated instruments — thermometers, metal detectors, checkweighers, refractometers. Without a central tracking system, certificates expire unnoticed. BRC Clause 6.2 requires documented evidence that all monitoring equipment is within calibration. A single expired certificate can trigger a major non-conformance.
02
Allergen Changeover Has Zero Margin for Incomplete Records
Moving from a nut-containing gummy to a nut-free hard candy on the same line requires documented verification at every stage. If even one line clearance signature is missing, BRC auditors treat it as a potential allergen cross-contamination event — a major finding that immediately threatens Grade AA status regardless of what the line actually looked like.
03
Preventive Maintenance Gaps Show Up as Compliance Gaps
Under BRC Global Standard Issue 9, preventive maintenance programs must be documented and executed on schedule. A 6-week gap in metal detector records — even if the detector was functioning perfectly — is treated as a system failure, not a scheduling oversight. Paper-based PM systems produce these gaps routinely.
04
Corrective Actions Are Documented Reactively, Not Systemically
BRC requires that every corrective action be documented, root-caused, and verified as effective. Most plants create the corrective action form. Very few link it back to the specific equipment record, the specific product batch, or the specific PM task that was missed. Auditors look for the chain — and when it's broken, it's a finding.
The Oxmaint Solution
How Oxmaint Rebuilt Their BRC Compliance Stack
01
Calibration Asset Registry
Every calibrated instrument registered in Oxmaint with calibration due dates, certificate storage, and automated alerts 30 and 60 days before expiry. No more spreadsheet — one dashboard, full visibility.
02
Digital Allergen Changeover Checklists
Line-specific changeover templates with mandatory sign-off fields. No batch can advance without supervisor verification completed in Oxmaint. Every step timestamped and tied to the production record.
03
Scheduled PM With Compliance Triggers
Metal detector checks, CCP equipment inspections, and sanitation verifications scheduled and tracked in Oxmaint. Overdue tasks escalate automatically — a gap cannot form without a visible alert in the system.
04
Corrective Action Workflow
When a non-conformance is logged, Oxmaint creates a linked corrective action work order with root cause fields, effectiveness verification steps, and close-out sign-off — giving auditors the complete chain they need.
05
Audit-Ready Export in Minutes
BRC auditors can request records for any asset, any date range, any checklist type. Oxmaint generates the complete package — work orders, calibration logs, changeover records — in a single export rather than a two-week manual retrieval process.
06
Mobile-First for Line Technicians
Technicians complete inspections on a mobile device at the equipment — not back at a desktop. Photos, readings, and sign-offs are captured at the point of work. No transcription errors, no missing data from delayed entry.
Before vs. After
Manual Compliance vs. Oxmaint-Powered BRC Readiness
| BRC Requirement |
Before Oxmaint |
With Oxmaint |
| Calibration tracking (Clause 6.2) |
Spreadsheet — certificates expired undetected |
Automated 30/60-day alerts per instrument |
| Allergen changeover records |
Paper binder — 37% of runs missing supervisor sign-off |
Digital mandatory sign-off — 100% completion enforced |
| PM schedule compliance |
Paper schedule — gaps formed without visibility |
Overdue alerts escalate automatically, no gaps possible |
| Corrective action documentation |
Standalone form — not linked to equipment or batch |
Linked work order with root cause and close-out chain |
| Audit record retrieval |
2-week manual search across binders and spreadsheets |
Full export in under 30 minutes per date range |
| BRC audit outcome |
Grade B — repeated major findings in documentation |
Grade AA — zero major non-conformances on first audit |
If your facility is hitting the same documentation wall every audit cycle, the fix is a system change — not more procedures. Start a free trial and map your BRC compliance requirements into Oxmaint today, or book a demo to see how other food manufacturers are achieving and maintaining Grade AA certification.
Grade AA
First audit result post-implementation
Up from Grade B on three consecutive prior audits
0
Major non-conformances on first Oxmaint audit
Previously averaging 4-6 majors per audit cycle
100%
Allergen changeover completion rate
Mandatory digital sign-off enforced at every line
14 days
Pre-audit overtime eliminated
Audit package now generated in under 30 minutes
FAQ
Frequently Asked Questions
Does Oxmaint support BRC Issue 9 specifically, or older versions of the standard?
Oxmaint's CMMS is built around documentation requirements that apply across BRC Global Standard issues — the platform handles calibration tracking, PM scheduling, corrective action workflows, and audit-ready record retrieval regardless of which issue version your site is certified under. For BRC Issue 9, specifically, Oxmaint supports the documented maintenance program requirements in Section 4.7, calibration requirements in Clause 6.2, and corrective action documentation in Clause 3.7. Your team configures inspection templates and PM schedules to match your site's specific BRC scope — the system doesn't prescribe a template, it captures whatever your HACCP team defines as the required evidence.
How does Oxmaint handle allergen changeover verification for multi-allergen confectionery lines?
Oxmaint supports multi-step allergen changeover checklists with role-specific sign-off requirements. For a line transitioning from a nut-containing product to a nut-free product, you configure a changeover template that requires: production supervisor sign-off at each cleaning stage, quality sign-off on visual inspection and ATP swab results, and a final line clearance confirmation before the next product can be released. Each field is mandatory — the checklist cannot be submitted in a completed state with missing fields. Every completed changeover record is timestamped and linked to the relevant production batch record, giving your BRC auditor the full chain they need without any manual assembly.
Can Oxmaint integrate with the temperature monitoring systems already installed in our candy plant?
Oxmaint integrates with IoT temperature monitoring systems and SCADA platforms via API. For confectionery applications — chocolate tempering line monitoring, enrober jacket temperature, cold storage — real-time sensor data can feed into Oxmaint, triggering automated work orders when set-points are breached. This means the temperature deviation is documented, assigned to a technician, and linked to a corrective action record automatically — without requiring a technician to manually create a work order after discovering the event. For BRC purposes, this creates a complete, timestamped deviation record that satisfies the documented corrective action requirement under Clause 3.7.
How long does it take to get a confectionery plant fully set up in Oxmaint?
Most confectionery facilities are operationally live in Oxmaint within 2-4 weeks. The implementation follows a structured sequence: asset registry build (all production equipment, CCP monitoring devices, and calibrated instruments), PM schedule configuration (frequencies, responsible parties, and documentation requirements per BRC clause), checklist template creation (allergen changeover, metal detector checks, sanitation verifications, and any other site-specific requirements), and team training via the mobile app. There are no heavy implementation fees or dedicated consultants required — Oxmaint is designed for in-house setup, and the support team is available throughout the process.
BRC Compliance — Powered by Oxmaint
Your Next BRC Audit Doesn't Have to Feel Like a Fire Drill
Oxmaint gives confectionery plants the calibration tracking, allergen changeover documentation, PM scheduling, and audit-ready record export they need to achieve and maintain BRC Grade AA — without two weeks of pre-audit overtime every cycle.