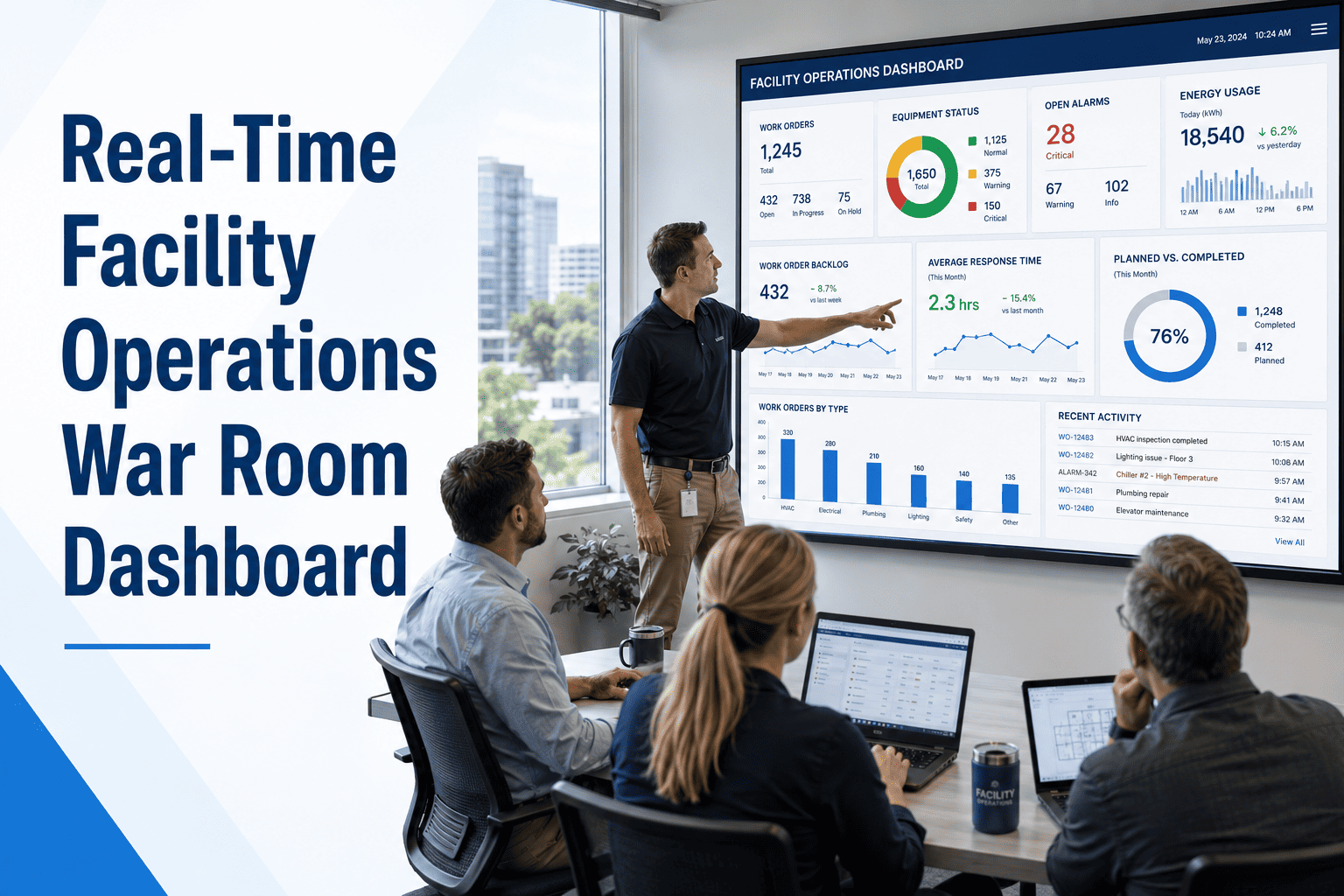
A 400-bed regional hospital received a Condition-level deficiency during an unannounced CMS survey when surveyors requested preventive maintenance records for ICU ventilators and the biomedical team needed two days to compile them from paper files. The resulting corrective action plan, legal review, and emergency CMMS implementation cost the facility over $340,000 — not counting the reputational damage and staff overtime. That hospital now runs every equipment inspection, life safety check, and compliance record through a centralized healthcare CMMS platform, producing audit-ready documentation within minutes of any surveyor request.
Facility Management for Healthcare: Joint Commission and CMS Compliance
Implement CMMS-driven compliance tracking for biomedical equipment, life safety documentation, and environment of care standards aligned with the 2026 Accreditation 360 restructuring.
Why Healthcare Facility Compliance Fails Without a CMMS
CMS surveyors arrive unannounced and request preventive maintenance logs, equipment inspection records, life safety documentation, and work order histories — expecting them immediately. Hospitals relying on spreadsheets, paper logs, or disconnected systems consistently fail this test. The Physical Environment standard at 42 CFR §482.41 is the single most cited deficiency area during hospital surveys, and reactive maintenance costs 4.8 times more than planned preventive maintenance. Sign up for Oxmaint to build the documentation infrastructure that answers every surveyor request with confidence.
Environment of Care Standards
The Joint Commission's Environment of Care chapter — now consolidated into the Physical Environment (PE) structure — requires hospitals to manage safety risks, hazardous materials, fire safety, medical equipment, and utility systems through documented programs with measurable outcomes.
Life Safety Code Compliance
NFPA 101 and NFPA 99 form the backbone of healthcare facility life safety requirements. CMS surveys verify fire door inspections, smoke/fire damper testing, fire alarm system maintenance, and egress path integrity — all requiring documented evidence available on demand. Book a demo to see how Oxmaint maps life safety ITM schedules to the new PE chapter codes.
Biomedical Equipment Management
The average hospital manages over 35,000 medical devices across 500 equipment categories. Each device requires specific maintenance schedules, calibration records, and traceable service histories producible on demand during Joint Commission surveys or FDA inspections. CMS requires all patient care equipment to be maintained in safe, reliable working order with documented PM completion rates.
Emergency Preparedness Documentation
CMS requires hospitals to maintain emergency preparedness programs including documented plans, policies, communication systems, and training exercises. Generator testing under NFPA 110, emergency water supply verification, and mass casualty drill records must be available for immediate surveyor review.
Stay Audit-Ready for Every Unannounced Survey
Oxmaint maps your maintenance workflows to the 2026 Joint Commission Physical Environment structure — so your documentation is survey-ready before surveyors arrive.
2026 Joint Commission Restructuring: What Changed
The Joint Commission's Accreditation 360 initiative, effective January 1, 2026, consolidated the Environment of Care (EC) and Life Safety (LS) chapters into a single Physical Environment (PE) chapter. While the underlying compliance work remains the same, how facilities must organize and report regulatory documentation during surveys has changed significantly. Hospitals using a CMMS built for healthcare compliance can remap existing procedures to the new PE codes without disrupting daily operations.
| Compliance Area | Previous Structure | 2026 PE Structure | CMMS Requirement |
|---|---|---|---|
| Fire Safety ITM | EC.02.03.05 | PE.04.01.01 | Automated NFPA schedule mapping |
| Medical Equipment PM | EC.02.04.01 | PE.03.01.01 | Risk-based PM with IFU documentation |
| Utility Systems | EC.02.05.01 | PE.05.01.01 | Generator and medical gas test logs |
| Hazardous Materials | EC.02.02.01 | PE.02.01.01 | SDS inventory with spill response records |
| Safety Management | EC.01.01.01 | PE.01.01.01 | Annual safety plans with outcome metrics |
How Oxmaint Supports Healthcare Compliance
Compliance Code Library
Pre-loaded Joint Commission PE codes, CMS K-Tags, and NFPA reference standards linked to every work order and inspection record for instant survey traceability.
Automated PM Scheduling
Risk-classified preventive maintenance calendars for biomedical equipment, life safety systems, and facility infrastructure with overdue escalation cascades.
Survey-Ready Reporting
Generate eBinder-ready compliance reports organized by PE chapter, asset category, and inspection date — producible within minutes of a surveyor's request.
Mobile Work Orders
Biomedical technicians complete inspections, log readings, and close work orders from the point of work — creating real-time compliance records with photo documentation.
Healthcare Compliance Regulatory Matrix
Hospital maintenance teams face simultaneous jurisdiction from five regulatory bodies. A unified CMMS consolidates documentation requirements across all agencies into a single audit-ready platform.
Joint Commission (TJC)
Risk-classified inventory, documented PM per manufacturer IFU, annual equipment management plan. Deficiency citations risk loss of accreditation.
CMS (42 CFR §482.41)
Life-safety compliance, emergency power testing, equipment maintenance for Medicare eligibility. Penalties up to $93K per day with 23-day termination timeline.
OSHA
Lockout/tagout compliance, hazard communication, bloodborne pathogen protection for biomed techs. Willful violations reach $161,323 per incident.
State Health Departments
Facility license standards, device maintenance per IFU, adverse event reporting, and water management program verification.
Consolidate Multi-Agency Compliance Into One Platform
Track Joint Commission, CMS, OSHA, and state requirements from a single dashboard with automated scheduling and instant audit documentation.
Frequently Asked Questions
Build Your Survey-Ready Compliance Infrastructure
Join hospitals that produce audit-ready documentation within minutes of any surveyor request — not after a two-day scramble through paper files.