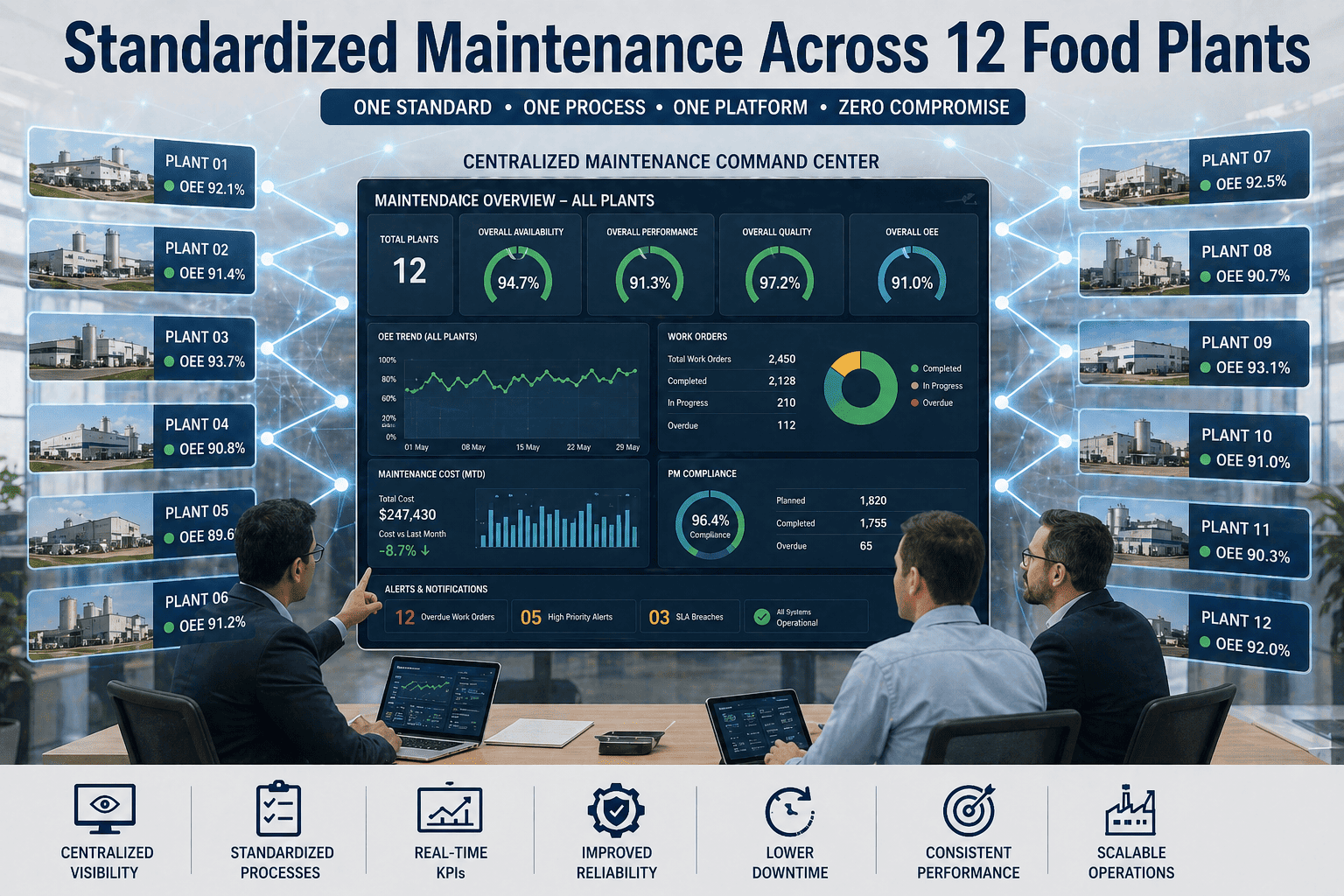
A dairy processing facility in Wisconsin achieved zero pathogen detections for 18 consecutive months after implementing a digital sanitation inspection workflow that replaced paper-based checklists with real-time verification and photographic evidence. Their previous approach relied on shift supervisors completing handwritten forms that were filed without review—allowing sanitation gaps to persist undetected until external audits revealed critical non-conformances. Now every cleaning task requires photographic verification, ATP swab results sync automatically to CMMS, and missed inspections trigger immediate alerts to quality managers. The facility passed their SQF certification audit with zero major findings and reduced cleaning chemical costs by 23% through optimized sanitizer concentration tracking. Sign up for Oxmaint to digitize your sanitation inspection workflows.
Sanitation Inspection Checklist for Food Manufacturing Facilities
Comprehensive sanitation verification ensures food safety compliance, prevents contamination events, and protects brand reputation. This checklist covers all critical control points from receiving docks to finished product storage with frequency guidelines and documentation requirements.
Why Sanitation Inspection Matters
Inadequate sanitation causes product recalls, regulatory penalties, and permanent brand damage. A structured inspection program catches contamination risks before they reach consumers. Book a demo to see how Oxmaint tracks sanitation compliance.
Food Contact Surfaces
Direct product contact equipment and utensilsFacility Environment
Floors, walls, drains, and overhead structuresEmployee Hygiene Stations
Handwashing, gowning, and transition areasStorage and Receiving
Raw materials, packaging, and finished goods areasChemical and Sanitizer Control
Storage, concentration, and application verificationPest Control Verification
Monitoring devices and exclusion measuresDigitize Your Sanitation Inspections
Oxmaint replaces paper checklists with mobile verification, photographic evidence, and automatic ATP result tracking—ensuring nothing gets missed.
Inspection Frequency Guidelines
Recommended verification frequencies based on contamination risk level and regulatory requirements. Sign up for Oxmaint to automate inspection scheduling.
| Inspection Area | Frequency | Verification Method | Documentation |
|---|---|---|---|
| Food Contact Surfaces | Each Shift | Visual + ATP Swab | Digital checklist + photo |
| Production Area Floors | Daily | Visual inspection | Digital checklist |
| Floor Drains | Weekly | Visual + swab sampling | Lab results + photo |
| Handwashing Stations | Each Shift | Supply check + temp verification | Digital checklist |
| Sanitizer Concentrations | Daily | Test strip verification | Concentration log |
| Pest Control Devices | Weekly | Visual inspection | Activity log + photo |
| HVAC Filters | Monthly | Visual + pressure differential | Maintenance record |
| Cold Storage Temps | Each Shift | Temperature log review | Continuous monitoring |
Oxmaint Sanitation Compliance Dashboard
Real-Time Verification TrackingPhoto Verification
Require photographic evidence for critical sanitation tasks to prove completion
ATP Result Tracking
Log swab results directly in CMMS with automatic threshold alerts for failures
Missed Inspection Alerts
Immediate notifications when scheduled sanitation checks are not completed on time
Pass Every Audit with Complete Documentation
Oxmaint maintains audit-ready sanitation records with timestamps, photos, and verification signatures that satisfy FDA, USDA, and third-party auditors.
Frequently Asked Questions
Prevent Contamination with Verified Sanitation
Digital sanitation workflows ensure every critical control point is verified, documented, and audit-ready. Oxmaint replaces paper checklists with real-time verification that catches sanitation gaps before they become contamination events.