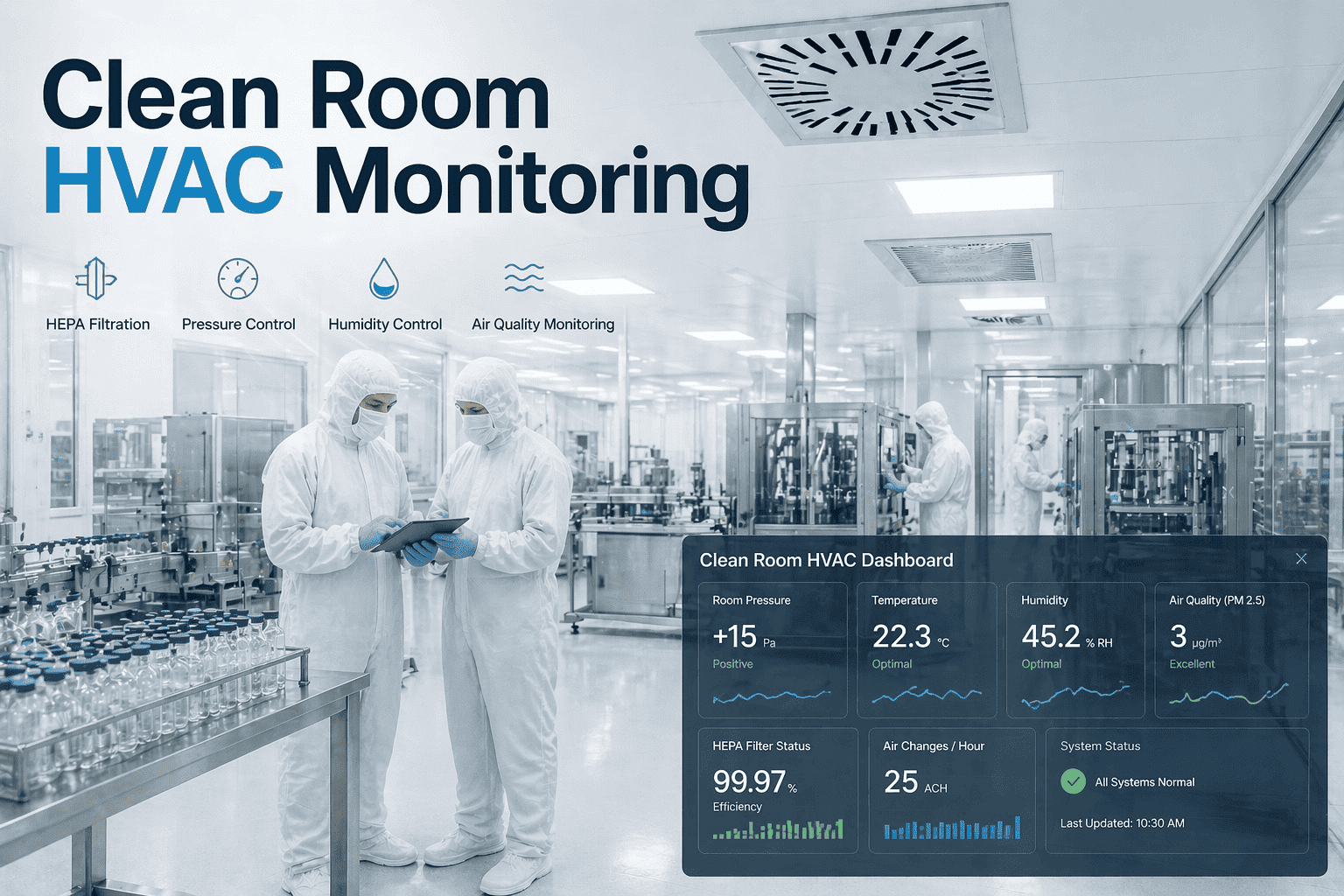
According to the global clean room monitoring market is projected to reach $7.2 billion by 2028, driven by pharmaceutical, semiconductor, and laboratory compliance requirements. However, data from regulatory audits indicates that 34% of clean room and laboratory HVAC systems fail at least one ISO 14644 or GMP parameter annually — with HEPA filter integrity (41% of failures), pressure differential (28%), and humidity stability (18%) representing the most common citation categories. Unlike standard commercial HVAC where gradual degradation triggers comfort complaints, clean room systems must maintain particle counts, pressure cascades, and temperature/humidity within narrow bands continuously, with deviation exceeding limits for any period risking product loss, batch rejection, or regulatory action. OxMaint's Clean Room Monitoring Module provides continuous tracking of HEPA filter pressure drop, room-to-room pressure differential, temperature stability, relative humidity, and particle count trends — generating compliance-ready reports and predictive work orders when any parameter approaches alert thresholds. Book a demo to see how quality managers are reducing clean room HVAC deviations by 52–68% within six months of deployment.
01
Clean Room and Laboratory HVAC Precision Environment Monitoring
HEPA integrity · Pressure differential · Humidity stability · ISO 14644 compliance · CMMS integration
34%Of clean room HVAC systems fail at least one ISO/GMP parameter annually
52–68%Reduction in clean room HVAC deviations with active monitoring
41%Of clean room failures attributed to HEPA filter integrity issues
Clean Room Compliance by the Numbers
$7.2B
Global clean room monitoring market value by 2028
CAGR 9.2% — driven by pharma and semiconductor sectors
ISO 5–8
Common clean room classifications per ISO 14644-1
Each class has defined particle count limits
0.05" w.g.
Minimum pressure differential required between clean and adjacent spaces
FDA GMP requirement for classified areas
Five Critical Clean Room Parameters That Require Continuous Monitoring
01
HEPA Filter Integrity & Pressure Drop
HEPA filters must maintain 99.97% efficiency at 0.3 microns. A 20% increase in pressure drop from clean baseline indicates filter loading — triggering replacement before airflow capacity drops below room classification requirements. A 15% decrease indicates filter media breach or seal failure, requiring immediate integrity testing. Monitoring detects both conditions 3–8 weeks before particle breakthrough.
02
Room-to-Room Pressure Differential
ISO 14644 and GMP require cascading pressure relationships to prevent contaminant migration. A laboratory at ISO 7 must maintain positive pressure relative to corridor (minimum 0.02–0.05 inches water column). Pressure loss of 30% from baseline triggers HVAC balancing or door seal inspection. Real-time differential monitoring prevents unreported cross-contamination events.
03
Temperature Stability (±1°C or tighter)
ISO 14644 specifies temperature control depending on process requirements — many pharma and semiconductor applications require ±1°C or better. Temperature drift beyond specified band triggers work orders for chilled water valve calibration, VAV box adjustment, or supply air temperature reset.
04
Relative Humidity Control
Many clean room applications require RH between 30–60% with ±5% control. Humidity excursions above 65% promote microbial growth; below 20% increases electrostatic discharge risk. Monitoring RH trends detects humidifier scaling, drain issues, or sensor drift — often 2–4 weeks before process impact.
05
Air Changes Per Hour (ACH) Verification
ISO classes specify minimum ACH: ISO 5 requires 350–500 ACH, ISO 6 requires 150–200 ACH, ISO 7 requires 40–60 ACH, ISO 8 requires 15–30 ACH. ACH reduction from design indicates filter loading, fan degradation, or duct leakage. Continuous monitoring of supply airflow validates classification maintenance between formal recertification cycles.
Clean Room HVAC Monitoring Checklist — Daily, Weekly & Monthly
01
HEPA Filter Pressure Drop — Daily Log
Record static pressure across each HEPA or ULPA filter bank. Compare to clean baseline. 20% increase triggers inspection; 15% decrease triggers integrity testing work order.
Prevents particle breakthrough
02
Room Pressure Differential — Continuous
Prevents cross-contamination
03
Temperature & RH — Continuous Recording
Maintain temperature within ±1°C of setpoint and RH within specified band (typically 30–60% ±5%). Excursions beyond thresholds require corrective action within 2 hours per GMP requirements.
Protects product integrity
04
Supply Airflow Volume — Weekly
Measure supply CFM to each terminal. Compare to design ACH requirements for room classification. Airflow drop of 15% from design initiates filter or fan investigation.
Maintains ISO classification
05
HEPA Integrity Scan — Semi-Annual / Annual
Perform aerosol photometer scan per ISO 14644-3. Any leak above 0.01% penetration requires filter replacement or repair. Document results with before/after certification.
Regulatory compliance
06
Particle Counts — Per ISO Class Schedule
ISO 5: every 6 months; ISO 6–7: every 12 months; ISO 8: every 24 months. Counts exceeding class limits trigger investigation and requalification before product release.
Validates clean room class
Monitoring Technology Comparison for Clean Room HVAC
| Monitoring Method | Parameters Monitored | Installation Complexity | Alerts & Workflow | Compliance Reporting |
|---|
| Manual / Paper Logs | Temperature, RH, pressure (spot checks) | None — technician rounds | No alerts — detection at next reading | Manual transcription — error prone |
| Building Management System (BAS) | Temp, RH, pressure, some airflow | High — requires full integration | Inside BMS only — no work order link | Requires export to separate reporting tool |
| Standalone Environmental Monitors | Temp, RH, differential pressure | Low — wireless or wired sensors | Email/text alerts — no work order automation | Limited — basic CSV export only |
| Periodic Third-Party Certification | Full ISO 14644 suite — particle counts, HEPA integrity | Service engagement | No monitoring between certifications | Full compliance report per visit |
| OxMaint Integrated Suite | HEPA pressure drop, pressure diff, temp, RH, ACH, particle trends | Low to moderate — wireless sensors + gateway | Auto work orders at configurable thresholds | Compliance-ready dashboard + export per ISO 14644 |
Source: OxMaint clean room and laboratory deployment data 2024–2025. Compliance reporting formats available for FDA, EMA, and ISO 14644 submissions.
ROI Impact at a Glance — Clean Room HVAC Monitoring
52–68%
Reduction in clean room environmental deviations
OxMaint customer data
$18K–45K
Average cost of product loss from single undetected HVAC deviation
Pharmaceutical batch rejection estimate
4–10 mo.
Payback period for monitoring hardware + software
Typical pharmaceutical / biotech clean room
“The clean rooms that pass regulatory audits without observation year after year are not the ones with the most expensive monitoring equipment — they are the ones that have closed the loop between environmental data and maintenance action. I’ve reviewed deviation reports from 60+ pharmaceutical and semiconductor clean rooms. The facilities that log HEPA pressure drops daily but only act when pressure drop exceeds a passive threshold still have filter failures that lead to particle excursions. The facilities that trend pressure drop over time, detect the rate of increase, and auto-generate filter replacement work orders when the slope predicts a breach within 90 days — those facilities don’t get deviations. OxMaint’s ability to trend clean room parameters continuously and trigger work orders based on rate-of-change, not just absolute thresholds, is precisely what transforms environmental monitoring from a compliance checkbox into a true predictive quality program.”
— Dr. James Okonkwo, PhD, PE · Clean Room Validation Specialist · 22 Years Pharmaceutical and Semiconductor Facility Compliance · ASHRAE Clean Space Committee Member
Stop relying on manual logs and mid-certification surprises. Start monitoring HEPA pressure, room pressure differential, and environmental stability continuously.
Frequently Asked Questions
What are the most common clean room HVAC monitoring parameters required for ISO 14644 compliance?
ISO 14644 does not mandate specific monitoring methods but requires documented proof that classified spaces maintain specified airborne particle concentration limits. The supporting parameters most auditors expect to see monitored include: HEPA/ULPA filter integrity (pressure drop or scan results), room pressure differential relative to adjacent spaces of different class, temperature (recorded at representative locations), relative humidity, air change rate or supply airflow volume, and particle counts at required intervals. For GMP-regulated facilities (pharmaceutical, medical device), FDA also expects continuous or frequent monitoring of pressure differential and critical environmental parameters with defined alert and action limits — typically recorded at least once per shift if not continuously.
Sign in to access OxMaint's ISO 14644 compliance reporting template.How often should HEPA filters in clean rooms be integrity tested?
ISO 14644-2 specifies that HEPA filter leak testing (integrity testing per ISO 14644-3) should be performed at maximum intervals of 24 months for ISO 5–7 clean rooms and 24 months for ISO 8, though many pharmaceutical facilities following GMP guidelines test annually or semi-annually depending on product risk. However, continuous monitoring of HEPA filter pressure drop is recommended between formal integrity tests. A stable pressure drop indicates filter integrity is maintained. A sudden decline of 15% or more from baseline suggests filter media breach or seal failure and should trigger immediate integrity testing regardless of scheduled interval. OxMaint tracks pressure drop trends and auto-generates integrity test work orders when deviation thresholds are exceeded.
Book a demo to see pressure-based integrity triggers.What is the difference between alert limits and action limits in clean room monitoring?
Per GMP and ISO 14644 guidance, alert limits are thresholds set at 50–80% of the specification limit. Crossing an alert limit does not require product action but does require investigation and increased monitoring frequency. Action limits are set at the specification limit (e.g., particle count limit for a given ISO class). Crossing an action limit requires immediate investigation, corrective action, and documented impact assessment on product exposed during the excursion. For example, in an ISO 7 clean room at rest (352,000 particles/m³ at 0.5 micron), an alert limit might be set at 250,000 particles/m³; action limit at 352,000 particles/m³. OxMaint allows separate configuration of alert and action thresholds per parameter, with work orders generated at alert level (investigation) and automatic escalation at action level (product hold notification).
Book a demo to configure tiered thresholds for your facility.Can OxMaint integrate with existing clean room monitoring hardware and sensors?
Yes. OxMaint's clean room module integrates with most common environmental monitoring hardware including: differential pressure transmitters (Dwyer, Setra, Honeywell), temperature/RH sensors (Vaisala, Rotronic, Siemens), particle counters (Lighthouse, Met One, TSI), and HEPA filter monitoring systems (AeroMed, Camfil, AAF). For facilities with existing BMS or BAS (Siemens Desigo, Johnson Controls Metasys, Schneider Electric), OxMaint can pull trend data via API or direct BACnet integration — avoiding duplicate sensors. For facilities without existing monitoring, OxMaint's wireless sensor suite (pressure, temp, RH, airflow) installs in 2–4 hours per clean room suite and transmits to a central gateway.
Start a free trial to map your existing sensor inventory. Ready to turn clean room monitoring from a compliance check into a predictive quality program?