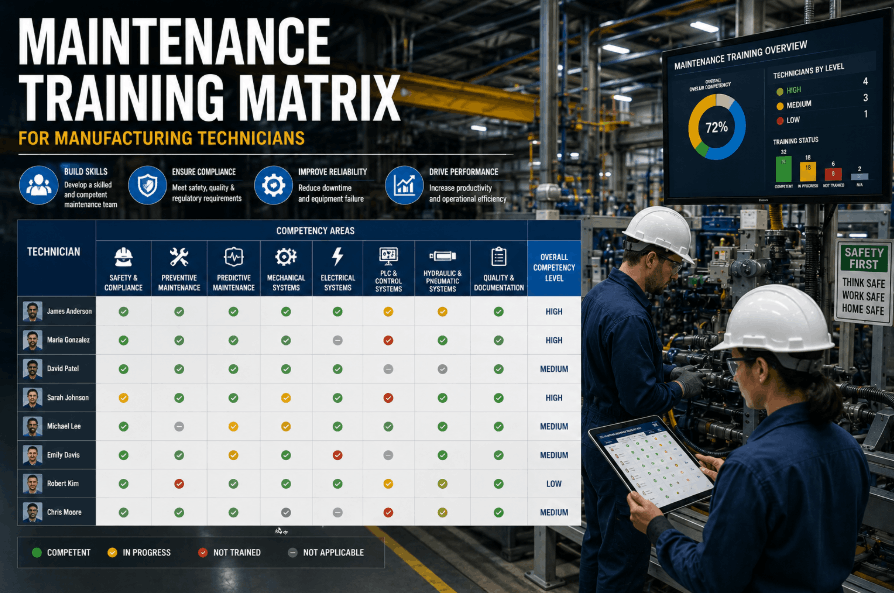
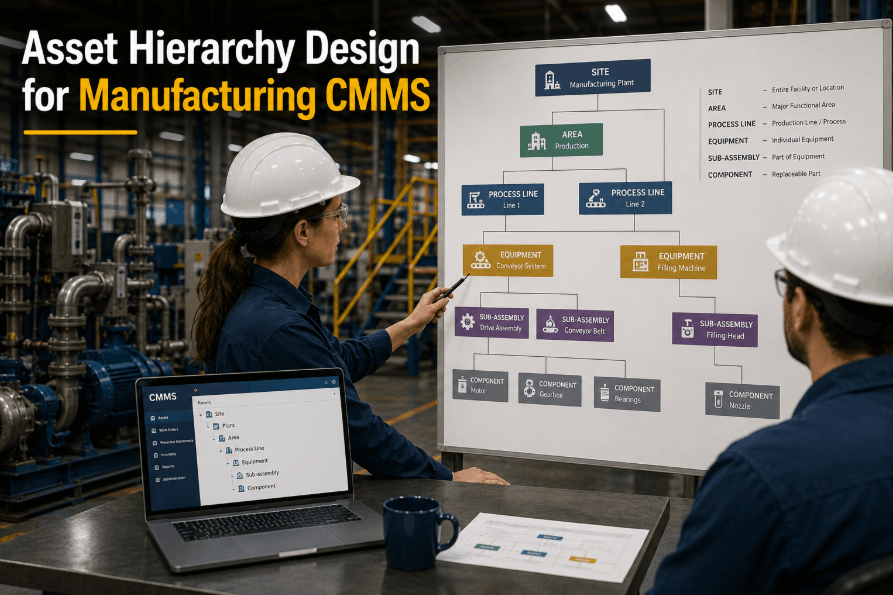
Pharmaceutical manufacturing operates under the strictest regulatory scrutiny of any industry — where a single undocumented equipment change can invalidate an entire batch worth millions, and calibration records missing a signature can halt production for weeks during FDA audits. In this environment, maintenance isn't just about preventing breakdowns — it's about maintaining validated states, documenting every intervention with audit-trail precision, and proving equipment performs exactly as qualified every single day. Facilities that consistently pass inspections don't just have good maintenance teams — they have pharmaceutical CMMS platforms that enforce GMP compliance at every work order, calibration cycle, and cleaning validation checkpoint. The difference between a warning letter and a successful audit often comes down to one question: can you instantly produce complete maintenance documentation for any asset in your facility?
Pharmaceutical · GMP Compliance · 2026
Pharmaceutical Manufacturing Maintenance: GMP & Validation
Comprehensive guide to maintaining pharmaceutical equipment under cGMP requirements — covering validation protocols, calibration management, cleaning validation, change control, and documentation standards that survive regulatory inspections.
$4.9M
average cost of FDA warning letter including remediation, batch losses, and reputation damage
63%
of pharma FDA observations cite inadequate equipment maintenance documentation
18mo
typical timeline from warning letter to full compliance restoration for maintenance gaps
100%
traceability required for all maintenance activities affecting validated equipment
Pharmaceutical facilities can't simply fix equipment and move on. Every maintenance action on validated systems requires documentation, impact assessment, revalidation consideration, and quality approval — creating compliance complexity that standard maintenance programs can't handle.
Equipment Validation Status
All critical equipment exists in validated states with Installation Qualification, Operational Qualification, and Performance Qualification documentation. Maintenance must preserve these validated conditions or trigger formal change control and revalidation.
Validation Critical
Calibration Traceability
Every measuring instrument requires calibration traceable to NIST standards with certificates, stickers, and scheduled recalibration dates. Out-of-calibration equipment invalidates any data it generated since last valid calibration.
Measurement Critical
Cleaning Validation
Equipment cleaning procedures are validated processes with documented limits for residue acceptance. Maintenance activities that affect cleanability require cleaning revalidation before production resume.
Quality Critical
Change Control Requirements
Replacing components, updating procedures, or modifying equipment configurations triggers formal change control with impact assessments, quality approvals, and documented justification for all changes.
Process Critical
Audit Trail Integrity
FDA requires complete maintenance records showing who did what, when, why, and with whose authorization. Paper systems with missing signatures or undated entries create audit findings that can escalate to consent decrees.
Compliance Critical
Preventive Maintenance Validation
PM procedures themselves require validation proving they don't adversely affect product quality. Changes to PM frequencies or tasks must be justified and approved through formal quality systems.
System Critical
IQ/OQ/PQ
Equipment Qualification Maintenance
Maintain equipment within qualified operating parameters. Document any maintenance that could affect validated performance. Conduct periodic requalification per validation master plan schedules.
Calibration
Instrument Calibration Program
Schedule calibrations based on risk assessment and manufacturer recommendations. Use NIST-traceable standards. Maintain calibration certificates and investigate out-of-tolerance findings with impact assessments.
Cleaning
Cleaning Validation Compliance
Follow validated cleaning procedures exactly. Document cleaning verification results. Assess whether maintenance work requires cleaning revalidation before returning equipment to production use.
PM Program
Preventive Maintenance SOPs
Execute PM tasks per validated procedures. Document completion with technician signatures and dates. Notify quality of any deviations from standard PM protocols for impact review.
Change Control
Maintenance Change Management
Assess all component replacements and procedure modifications for GMP impact. Route significant changes through formal change control with quality approval before implementation.
Documentation
Electronic Batch Record Integration
Link maintenance activities to affected batches. Enable quality teams to trace which batches ran on equipment before and after maintenance interventions for investigation purposes.
Every maintenance intervention follows a strict pathway ensuring validated status preservation, documentation completeness, and quality oversight at critical decision points.
Step 1
Maintenance Request & Assessment
Technician or operator identifies equipment issue. Request routed to maintenance with equipment ID, problem description, and criticality level for validated systems.
→
Step 2
Impact Analysis & Approval
Maintenance reviews whether work affects validated parameters. Critical equipment changes require quality review and formal authorization before work proceeds.
→
Step 3
Work Execution & Documentation
Technician performs maintenance following SOPs. Documents parts replaced, procedures followed, and any deviations encountered during repair or PM activity.
→
Step 4
Verification & Quality Release
Equipment tested to confirm functionality. Quality reviews documentation for completeness. Formal release to production issued only after all records verified.
| Instrument Type |
Typical Cal Frequency |
Traceability Requirement |
Documentation |
| Temperature Probes |
Annually |
NIST-traceable thermometer |
Certificate with as-found/as-left data |
| Analytical Balances |
Quarterly |
Certified mass standards |
Calibration certificate with uncertainty |
| Pressure Gauges |
Semi-Annually |
Calibrated pressure source |
Multi-point verification records |
| HVAC Differential Pressure |
Quarterly |
Traceable manometer |
Room-specific cal records with trends |
| pH Meters |
Monthly |
Buffer solution certificates |
Two-point cal with slope verification |
| HPLC Systems |
Per SOP Schedule |
Manufacturer qualification |
System suitability and performance quals |
All calibrations require unique identification labels, next due dates, and maintained certificates accessible during audits.
01
Automatic Audit Trails
Every work order, calibration, and PM completion creates timestamped records showing technician ID, completion date, and any approvals required — eliminating handwritten logs prone to errors.
02
Calibration Due Date Alerts
System tracks every instrument calibration schedule and sends automatic alerts before expiration — preventing use of out-of-cal equipment that invalidates production batches.
03
Electronic Signature Compliance
21 CFR Part 11 compliant electronic signatures with unique user IDs and password controls — meeting FDA requirements for electronic record authenticity and integrity.
04
Validation Document Management
Store IQ/OQ/PQ protocols, validation reports, and change control documentation directly linked to equipment assets — instant retrieval during audits without searching filing cabinets.
05
Change Control Integration
Route equipment modifications through formal approval workflows with quality reviews, impact assessments, and documented justifications all tracked within one platform.
06
PM Procedure Version Control
Maintain historical versions of maintenance procedures with effective dates and change reasons — proving procedures followed were current and approved at time of execution.
Must Have Available
Complete equipment inventory with unique IDs and validation status
PM schedules with actual completion dates vs planned dates
Calibration certificates for all measuring instruments with due dates
Work order history showing all maintenance performed on critical equipment
Change control records for any equipment modifications or procedure updates
Technician training records proving competency for tasks performed
Vendor qualification documentation for contract maintenance services
Spare parts traceability showing lot numbers and certificates of analysis
Common Inspection Findings
Incomplete or missing signatures on maintenance records
Calibration certificates without as-found/as-left data documented
PM tasks completed late without documented justification or impact review
Equipment modifications made without formal change control approval
Maintenance procedures not reflecting actual practices observed on floor
Missing documentation proving cleaning validation after major repairs
Inadequate equipment identification labels or calibration stickers
Lack of batch traceability for maintenance performed during campaigns
94%
reduction in maintenance-related audit observations after implementing pharmaceutical CMMS
8hrs
average time saved per FDA inspection through instant electronic record retrieval vs paper files
100%
PM completion rate achievable with automated scheduling and escalation workflows
Maintain Pharmaceutical Equipment the GMP-Compliant Way
Oxmaint provides pharmaceutical-grade CMMS with 21 CFR Part 11 compliance, calibration tracking, validation document management, and audit-ready maintenance records that survive even the most rigorous FDA inspections. Built specifically for regulated industries where documentation quality matters as much as equipment reliability.
What is GMP compliance in pharmaceutical manufacturing maintenance?
GMP (Good Manufacturing Practice) compliance requires maintaining equipment according to validated procedures, documenting all maintenance activities with complete traceability, and ensuring work doesn't compromise product quality or equipment validation status. It means every maintenance action must be planned, approved, executed per SOP, and documented with audit trail integrity.
How often must pharmaceutical equipment be calibrated?
Calibration frequency depends on risk assessment, criticality to product quality, and manufacturer recommendations. Critical instruments like analytical balances may require quarterly calibration, while temperature probes might be annual.
Pharmaceutical CMMS platforms auto-schedule calibrations and prevent overdue equipment use.
What happens if maintenance documentation is incomplete during FDA inspection?
Incomplete records generate Form 483 observations that can escalate to warning letters if patterns emerge. Missing signatures, undated entries, or gaps in calibration records force you to prove affected batches remain acceptable — potentially resulting in batch rejections, recalls, or consent decrees requiring third-party oversight.
How does change control apply to equipment maintenance?
Replacing components with different part numbers, modifying procedures, or changing PM frequencies all require change control evaluation. Quality must assess whether changes affect validated status and approve before implementation.
Book a demo to see how Oxmaint automates change control workflows for maintenance activities.
Protect Your Manufacturing License with Compliant Maintenance
Join pharmaceutical manufacturers using Oxmaint to maintain validated equipment states, track calibrations automatically, generate audit-ready documentation, and ensure every maintenance activity meets cGMP requirements. Start your free trial today — no credit card required.